Need help choosing the right product?
Our tool will match the best product to your needs
Launch product selectorWelcome to the Global Website
We have detected that you may prefer the Global site. Please use the language dropdown above to change your selection if required.
A tablet is a pharmaceutical dosage form. It comprises a mixture of active substances and excipients, usually in powder form, compressed or compacted into a solid. Coating is the last unit operation where a tablet’s external features can be modified. This process is used for multiple reasons:
Tablet coating is the process of applying a suspension or solution containing the components needed for the coating formation, on a moving bed of tablets, keeping a constant temperature in the bed. It takes place within a controlled atmosphere inside a perforated rotating drum. Angled baffles fitted into the drum and air-flow inside the drum provide means of mixing the tablet bed. As a result, the tablets are lifted and turned from the sides into the center of the drum, exposing each tablet surface to an even amount of deposited/sprayed coating.
The liquid spray coating is then dried onto the tablets by heated air blown through the tablet bed from an inlet fan. The air-flow is regulated for temperature and volume to provide controlled drying and extracting rates, and at the same time, maintaining a slightly negative drum pressure relative to the room to provide an isolated process atmosphere.
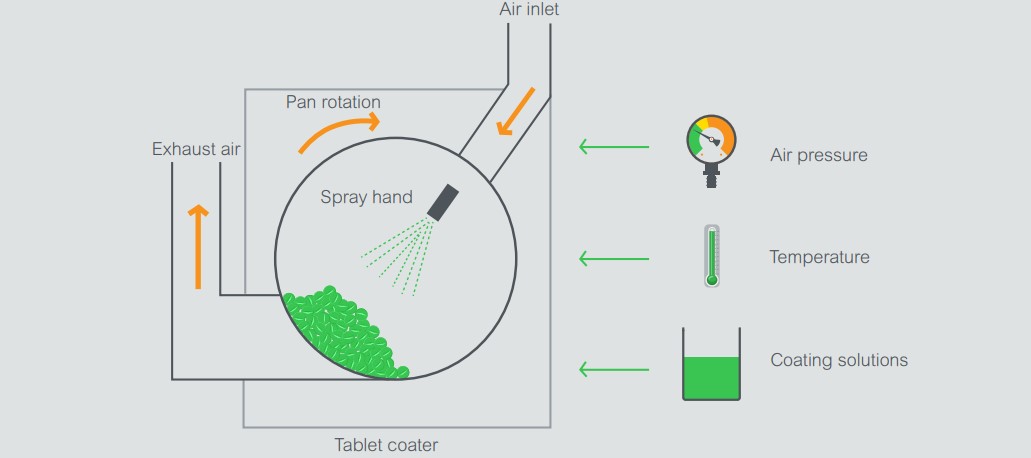
Non-uniform coating created by inefficient mixing is one of the main issues that can occur during coating. There are several variables to consider:
The coating process is usually a batch driven task consisting of the following phases:
As an automation and information technology leader our solutions meet the Electronic Record and Electronic Signature requirements as defined by the US and EU regulatory bodies.
To make sound decisions, you need to trust your data. Key regulatory bodies (FDA, EMA, WHO) and some advisory bodies (PIC/S, ISPE) have agreed on the Data Integrity related ALCOA+ concept. ALCOA+ defines that data should be Attributable, Legible, Contemporaneous, Original, and Accurate + Complete, Consistent, Enduring, and Available. As an experienced solution supplier, well established in life science processes, Eurotherm is a main supporter of that vision and contributed to the definition and the revision of some of these guidelines.
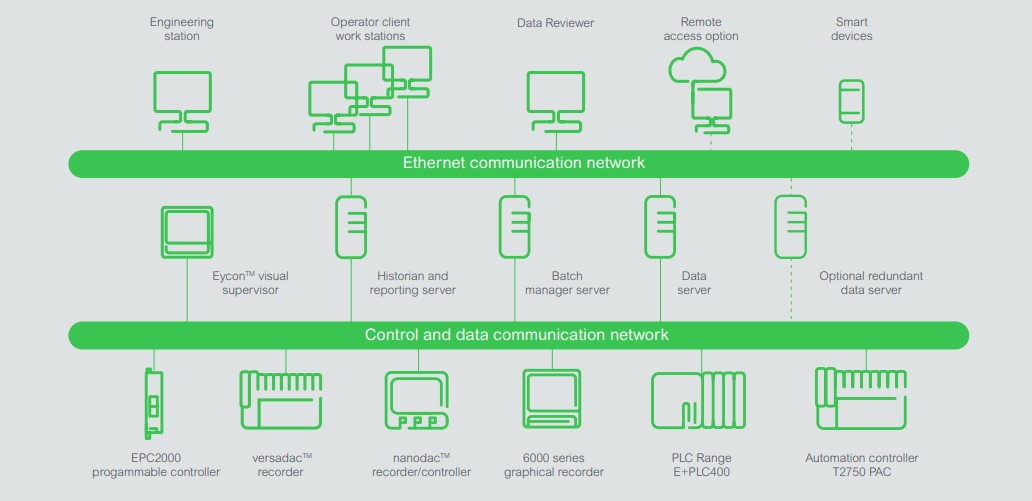
Business investments should be future-proof and audits hassle free. Eurotherm has developed and widely applied a set of good engineering practise (GEP) qualification documents based on ISPE GAMP 5 guidelines to assist with achieving these goals. Qualification documents can be maintained in electronic format. Industry is progressing from manual standard operating procedure (SOP) based manufacturing operations to a digitalized paperless quality systems approach based on FDA and ICH guidelines.
In a quality by design (QbD) approach, product quality is continuously monitored and controlled at the earliest stages, rather than waiting for a process to end. Pharmaceutical manufacturers need to focus on identifying, controlling, and validating process variables that could cause a non-compliant result. This is accomplished by managing the quality target product profile (QTPP), the critical quality attributes (CQA), and the critical process parameters (CPP). As defined by the process analytical technology (PAT) approach, Eurotherm can assist with measurement and performance analytics on CQAs and help manage CPP deviations, providing time-stamped evidence for correlation of parameter behaviors at their occurrence.
Eurotherm control and data recording solutions are EcoStruxure-ready, providing a Data Integrity layer within the Schneider Electric EcoStruxureTM platform. EcoStruxure is Schneider’s open IoT-enabled system architecture aiding the digital transformation to Pharma 4.0 technology.
Product Selector
Our tool will match the best product to your needs
Launch product selectorContact Us