Välkommen till Sweden-webbplats
Vi har upptäckt att du kanske föredrar Global-webbplatsen. Använd språkmenyn ovan för att ändra ditt val om det behövs
Go to Global
Välkommen till Sweden-webbplats
Vi har upptäckt att du kanske föredrar Global-webbplatsen. Använd språkmenyn ovan för att ändra ditt val om det behövs
Eurotherm has decades of knowledge and understanding of the requirements for capturing, storing and retrieving electronic records and will ensure your data is kept safe at every stage of its life.
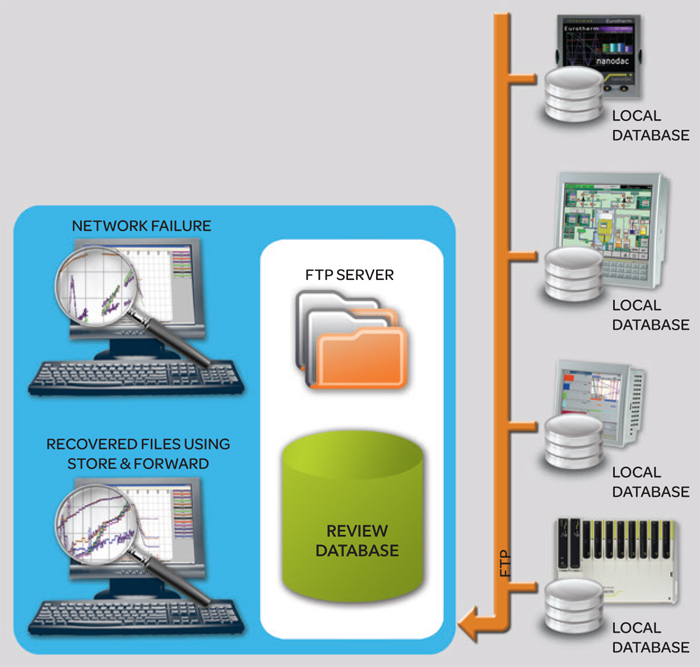 The files created in the versadac recorder can be archived to numerous locations to best suit operational requirements and add efficiency to the process. File transfer protocol (FTP) is used to send files via Ethernet at configurable intervals to an FTP server. A second FTP server address can also be set for backup in case the primary server is not available. The PC Review software further automatically builds a database of recorded files over the network. For the ultimate in efficiency in handling recorded data, the EcoStruxure Manufacturing Compliance Advisor Director application can be used to store files on a fully secure and managed datahandling facility – enabling user access from any web-enabled device anywhere.
The files created in the versadac recorder can be archived to numerous locations to best suit operational requirements and add efficiency to the process. File transfer protocol (FTP) is used to send files via Ethernet at configurable intervals to an FTP server. A second FTP server address can also be set for backup in case the primary server is not available. The PC Review software further automatically builds a database of recorded files over the network. For the ultimate in efficiency in handling recorded data, the EcoStruxure Manufacturing Compliance Advisor Director application can be used to store files on a fully secure and managed datahandling facility – enabling user access from any web-enabled device anywhere.
One of the many unique features provided by Eurotherm archiving strategies is the “Store & Forward” functionality. This is a selfhealing, validated archiving system that automatically stores data in the measuring device during a communications failure and, once communications have been restored, pulls the missing data into the filing system, ensuring complete and total data integrity.
All services that can access recorded data come with comprehensive user access security – from the view of the live versadac recorder through to access to EcoStruxure Manufacturing Compliance Advisor Director which may hold decades worth of records.
User access to a versadac instrument
Access to the versadac recorder is via the PC iTools configuration software. Each user can be given a unique user name and password with specific access rights. This not only ensures protection of privileged areas but also simplifies operation as dynamic menus only show relevant options.
Electronic Signatures
The versadac scalable recorder supports the use of Electronic Signatures in accordance with FDA 21 CFR Part 11. All user actions can be configured to require signing or signing and authorization. The ability to sign and authorize is configured on a user by user basis and incorporates signature control elements including account retirement.
Each user account has a completely unique username and password. Passwords can be controlled with defined expiry and configurable minimum length. Further protection is enabled with automatic timed logoff along with automatic disabling of accounts and notification on failed login attempts.
Security Manager software is available to provide a process wide solution to security management with user IDs, passwords and access permissions maintained in a single database.
The versadac recorder offers sample and recording rates of up to 8Hz with high accuracy inputs. Data can also be recorded over comms channels as well as from the outputs of mathematical functions and application blocks. Any metadata and audit trail information is also securely recorded along with the process information.
All data captured is stored in an impressive 100MB internal flash memory which provides constant recording of process conditions. For applications within industries such as pharmaceutical and heat treatment, recorded files can be in secure, tamper resistant, binary, check-summed format (known as “UHH” files). If appropriate to the application, files can also be created in open CSV format for direct use in programmes such as Microsoft® Excel®.
Review software and EcoStruxure Manufacturing Compliance Advisor Director offer powerful search capabilities which are specifically designed to quickly find and retrieve process data. Searches can be carried out by filename, date, time or batch name/number. Once the correct file is located, the user can quickly recreate the view of the process with Review software or generate a customised report using Dream Reports.
Contact Us