Need help choosing the right product?
Our tool will match the best product to your needs
Launch product selectorWelcome to the Global Website
We have detected that you may prefer the Global site. Please use the language dropdown above to change your selection if required.
Suspended or dissolved particles, organic compounds, impurities and other contaminants prohibit the usage of tap water in laboratory applications and scientific research. Parameters such as resistivity, conductivity, size of particulate matter and concentration of microorganisms are used to categorize water quality and, therefore, specify intended uses for water. Some applications can tolerate the presence of specific impurities in the water, but others, such as High Performance Liquid Chromatography (HPLC) require removal of the majority of contaminants.
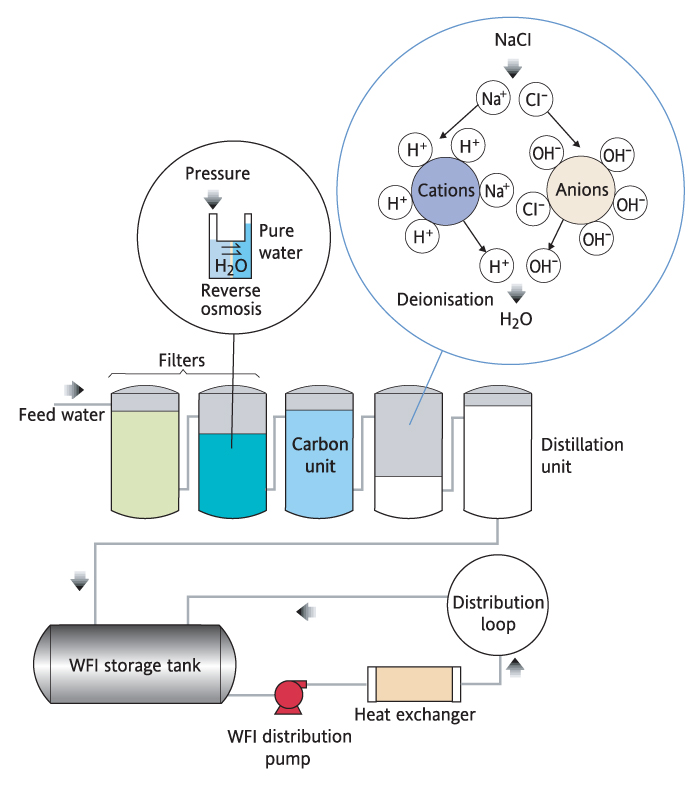
Water is an excellent solvent and can be sourced from almost anywhere on Earth. This property makes it prone to all kinds of contamination.
Scientific applications require elimination of certain types of contaminants. On the other hand, pharmaceutical productions require, in most cases, near-total removal of impurities (criteria dictated by specific standards or local/international regulatory bodies).
There are a number of methods commonly used to purify water. Their effectiveness is linked to the type of contaminant being treated and the type of application the water will be used for.
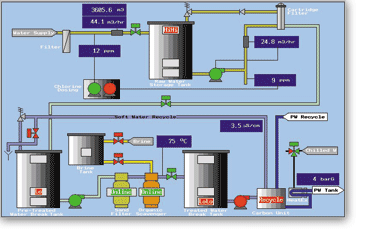
Sanitization of water purification equipment with hot water is achieved via an appropriate combination of exposure time and temperature. A primary use for this process is to deactivate viable microbes. It is worth mentioning that Endotoxin reduction is not achieved as a direct result of the hot water sanitization process.
Based on the feed water source, system operating conditions and the end-user’s operating and maintenance procedures, traditional chemical cleaning processes may still be required.
Sanitization using hot water involves incorporating heat exchangers into the traditional clean in place (CIP) system to gradually heat and cool water circulating through the reverse osmosis membrane system. Membrane manufacturers commonly stipulate a controlled heating and cooling rate to protect against irreversible damage to the membrane and ensure the system’s long-term performance.
A typical hot water sanitization sequence would consist of the following phases:
A control system must therefore provide flexibility in the way in which accurate and repeatable control of the sterilization is achieved and will include the following features:
The EyconTM Visual Supervisor is an ideal solution for this application.
Product Selector
Our tool will match the best product to your needs
Launch product selectorContact Us