Need help choosing the right product?
Our tool will match the best product to your needs
Launch product selectorWelcome to the Singapore Website
We have detected that you may prefer the Global site. Please use the language dropdown above to change your selection if required.
Global agencies such as the WHO (World Health Organization), FDA (Food and Drug Administration), EMA (European Medicine Agency), and associations such as PIC/S (Pharmaceutical Inspection Co-operation Scheme), and the ISPE (International Society for Pharmaceutical Engineering), have established a consensus on Data Integrity guidance to manage the quality of electronic records in the Life Science industry.
Data Integrity is an important part of any production quality requirement on the GMP (Good Manufacturing Practice) environment.
Therefore, in regard to the ALCOA+ concept argument “ACCURATE” and “CONSISTENT” loss of any data resulting in gaps in the historic information is not acceptable for any production batch and any measurement on the Environmental Monitoring System.
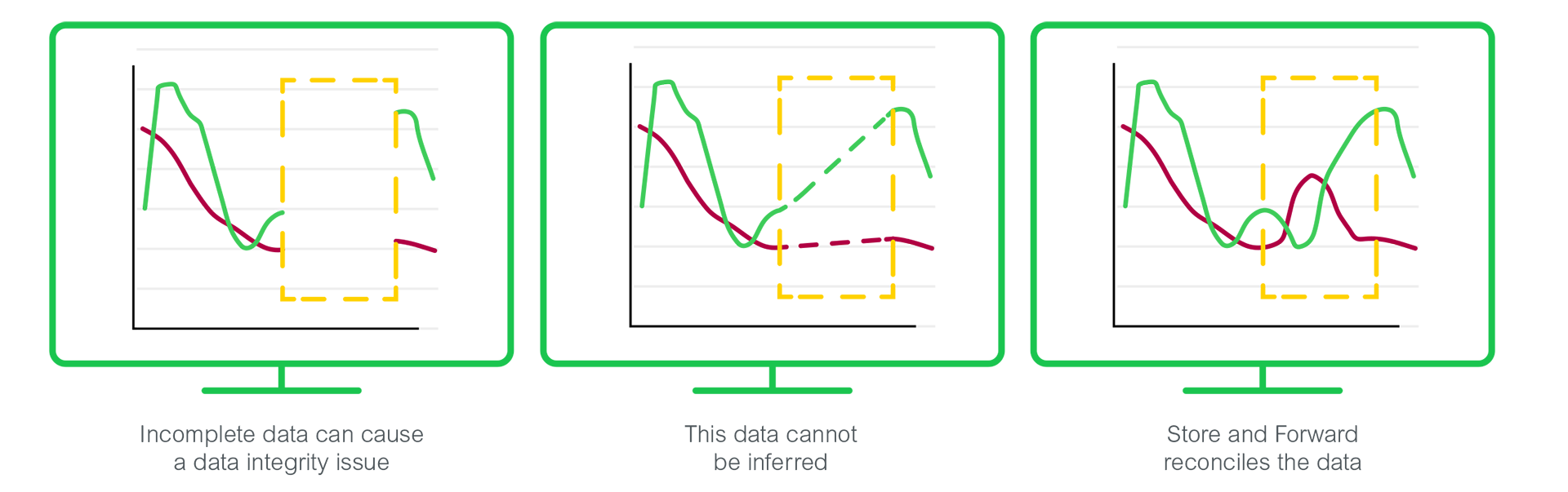
Eurotherm recording products record data at the point of measurement for archiving later, reducing the risk of data loss if the server or communications are temporarily lost. When used in harmony with a historian server, the Eurotherm Store and Forward feature aids reliability of archiving, by reconciling any missing data to storage databases when communications are resumed. This supports the ALCOA+ data integrity concept by providing original, attributable data that is contemporaneous, consistent and complete.
[gdpr-youtube id=”nYvvWTZNWpw
Typically, long-term historian data is written to a computer server, but loss of communications with a measuring device can cause a loss of data in production batch or on the Environmental Monitoring System.
The concept of Store and Forward fills these gaps in the collected data by using the Historical Raw Data and Audit Trail stored locally on a measuring device.
The local historical Raw Data, Meta-Data and Audit trail are forwarded to the Historian server and used to replace any gaps in collected data by extracting data contained in the Tamper Resistant recording (.uhh) files and writing it to the database.
Product Selector
Our tool will match the best product to your needs
Launch product selectorContact Us